Your brain uses 20% of your body’s energy, even though it’s only 2% of your weight. This energy comes from mitochondria – tiny structures in your cells. When mitochondria don’t work well, the brain struggles, which may contribute to psychiatric conditions like depression, bipolar disorder, anxiety, ADHD, and schizophrenia.
Key Insights:
- Mitochondrial Dysfunction: Linked to energy deficits, oxidative stress, and impaired brain function.
- Psychiatric Disorders: Depression shows reduced energy in the prefrontal cortex; bipolar disorder shifts to less efficient energy use; schizophrenia involves mitochondrial abnormalities.
- Stress Impact: Chronic stress damages mitochondria, worsening mental health.
- Various treatments, including nutrients like CoQ10 (e.g., CoQ10, Omega-3s, B12), exercise, sleep, and stress management improve mitochondrial function.
Testing & Solutions:
- Biomarkers like lactate, mtDNA, and oxidative stress indicators can diagnose dysfunction.
- Functional lab tests and neuroimaging help pinpoint issues.
- Treatments include tailored supplements, lifestyle changes, and emerging therapies like mitochondrial-targeted antioxidants and NAD⁺ boosters.
Addressing mitochondrial health offers a new perspective on mental health treatment, focusing on energy production and cellular repair.
How Mitochondria Shape Your Mind, Mood, & Mental Health with Dr. Martin Picard
Mitochondrial Dysfunction and Psychiatric Disorders
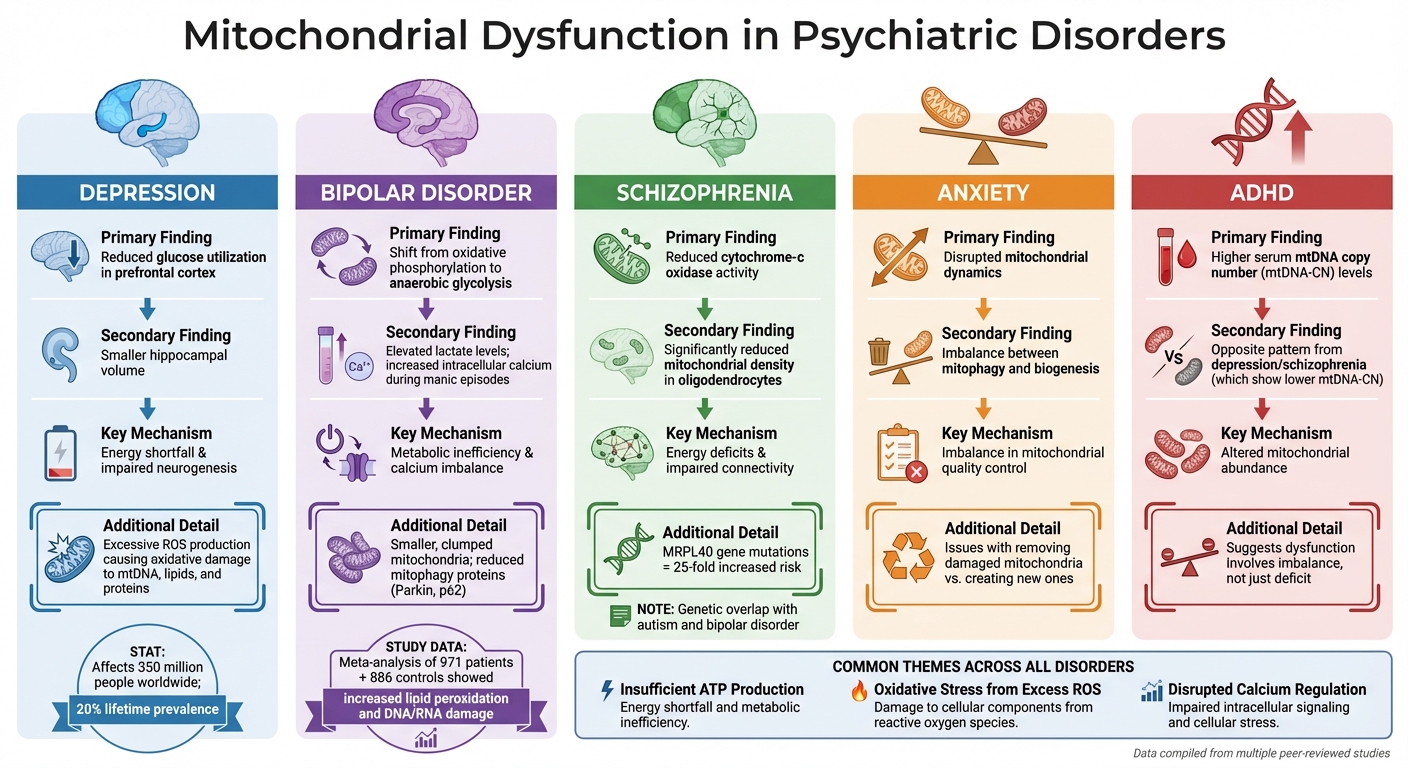
Mitochondrial Dysfunction Across Major Psychiatric Disorders
Mitochondrial dysfunction plays a role in a variety of psychiatric conditions. Depression alone impacts over 350 million people worldwide, with around 20% experiencing it at some point in their lives.
“Mitochondrial dysfunction not only hampers cells to meet energy requirement but might also be involved in the impairment of neuronal communication and cellular resilience, which prop up mood disorders and psychotic disorders.” – Yashika Bansal, Pharmacology Research Laboratory, Panjab University
The brain’s energy demands are extraordinary, making it especially vulnerable to mitochondrial issues. For instance, a single cortical neuron uses billions of ATP molecules every second. When mitochondria fail to meet these energy needs, neurotransmitter production and the development of new neural connections can be disrupted. Below, we explore how these disruptions manifest in specific psychiatric conditions.
Depression and Mitochondrial Function
In depression, mitochondrial dysfunction creates an energy crisis that affects critical areas of the brain. Studies show that individuals with depression often exhibit reduced glucose utilization in the prefrontal cortex and a smaller hippocampal volume.
Beyond energy production, malfunctioning mitochondria produce excessive reactive oxygen species (ROS), leading to oxidative damage to mitochondrial DNA, lipids, and proteins. This oxidative damage worsens mitochondrial performance, particularly in the hippocampus – a region essential for memory and mood regulation. Proper neuronal differentiation in this area relies on mitochondrial biogenesis.
Chronic stress exacerbates these problems via the HPA axis. Elevated glucocorticoid levels can impair the respiratory chain, increase ROS production, and cause structural changes in hippocampal neurons. This connection helps explain why prolonged stress is a major risk factor for depression.
Bipolar Disorder and Mitochondrial Function
Bipolar disorder is associated with a shift in energy metabolism, moving from oxidative phosphorylation to less efficient anaerobic glycolysis. This shift is marked by elevated lactate levels in the frontal cortex and cingulate regions, often alongside increased intracellular calcium during manic episodes.
Structural abnormalities are also evident. Bipolar patients often have smaller, clumped mitochondria. A meta-analysis involving 971 bipolar patients and 886 controls identified significant increases in markers of lipid peroxidation and DNA/RNA damage.
Additionally, a reduction in mitophagy-related proteins like Parkin and p62 means damaged mitochondria are not effectively cleared, leading to increased cell death. Mutations in the ANT1 gene, which is involved in ATP transport, further contribute to serotonin turnover and the hyperactivity observed in bipolar disorder.
Mitochondrial Dysfunction in Schizophrenia
Schizophrenia is characterized by pronounced mitochondrial abnormalities. Postmortem studies reveal reduced cytochrome-c oxidase activity in key brain areas . Oligodendrocytes in the prefrontal cortex, which insulate nerve fibers, show significantly reduced mitochondrial density. Genetic studies also highlight this connection: mutations in the MRPL40 gene, which encodes a mitochondrial ribosome component, are linked to a 25-fold increased risk of schizophrenia.
Moreover, genetic overlap between autism, schizophrenia, and bipolar disorder suggests shared mitochondrial risk factors.
Anxiety, and ADHD
Anxiety and ADHD show different mitochondrial patterns. While disorders like schizophrenia and depression are associated with lower mitochondrial DNA copy numbers (mtDNA-CN), ADHD and autism are linked to higher mtDNA-CN levels in serum.
This indicates that mitochondrial dysfunction may involve not just deficits or excesses, but a broader imbalance in mitochondrial regulation. In anxiety, research suggests that disruptions in mitochondrial dynamics – specifically the balance between mitophagy (removing damaged mitochondria) and biogenesis (creating new mitochondria) – play a role in its development.
| Disorder | Primary Mitochondrial Finding | Key Mechanism |
|---|---|---|
| Depression | Reduced glucose utilization; decreased hippocampal volume | Energy shortfall & impaired neurogenesis |
| Bipolar Disorder | Shift to anaerobic metabolism; increased lactate; high intracellular calcium | Metabolic inefficiency & calcium imbalance |
| Schizophrenia | Reduced cytochrome-c oxidase activity; lower mitochondrial density in oligodendrocytes | Energy deficits & impaired connectivity |
| Anxiety | Disrupted mitophagy and biogenesis | Imbalance in mitochondrial dynamics |
| ADHD | Higher serum mtDNA-CN levels | Altered mitochondrial abundance |
Despite the differences across these conditions, recurring themes emerge: insufficient ATP production, oxidative stress from excess ROS, and disrupted calcium regulation. Targeting mitochondrial health could offer new pathways for addressing a range of psychiatric disorders.
Testing for Mitochondrial Health
Diagnosing mitochondrial dysfunction in psychiatric patients requires specific lab tests that evaluate how effectively these cellular energy centers are working. Even small disruptions in mitochondrial function can have a noticeable impact on mental health. Testing allows clinicians to identify where energy production is faltering and design precise treatment strategies.
Key Biomarkers for Mitochondrial Function
Various biomarkers can shed light on mitochondrial health. Metabolic markers like lactate, pyruvate, and the lactate-to-pyruvate (L:P) ratio help determine if cells are relying on less efficient anaerobic glycolysis. For example, a blood lactate level exceeding 2.4 mmol/L or an L:P ratio over 20–25 often signals this metabolic shift.
Tests measuring enzymatic activity provide direct insights into mitochondrial energy production. These tests assess the function of respiratory chain complexes (I, II, and IV) and Citrate Synthase. One study found that patients with Major Depressive Disorder who responded to SSRI treatment had higher baseline Citrate Synthase levels and mitochondrial DNA copy numbers. Interestingly, reductions in Complex I activity during treatment were linked to lower depression severity.
Oxidative stress markers, such as malondialdehyde (MDA) for lipid damage and antioxidant enzymes like superoxide dismutase (SOD), catalase, and glutathione (GSH/GSSG), measure the body’s ability to manage reactive oxygen species. A meta-analysis involving 971 bipolar patients revealed increased lipid peroxidation and DNA/RNA damage compared to controls.
Protein biomarkers
Protein biomarkers are gaining recognition for their sensitivity. Growth Differentiation Factor 15 (GDF-15) and Fibroblast Growth Factor 21 (FGF-21), known as “mitokines”, signal mitochondrial distress. GDF-15 has shown impressive diagnostic accuracy, with an Area Under the Curve (AUC) reaching 0.997. Clinical thresholds have been established: GDF-15 levels above 710.0 pg/mL and FGF-21 levels in the 200–350 pg/mL range. FGF-21 demonstrated 82.8% sensitivity and 91.7% specificity in pediatric studies.
“In large biomarker comparison studies, GDF-15 followed by FGF-21, seem to have the greatest value though they are still not perfect.” – Brian J. Shayota, MD
Genetic markers, including mitochondrial DNA copy number (mtDNAcn) and circulating cell-free mtDNA (ccf-mtDNA), provide further insight into mitochondrial density and cellular stress.
Neuroimaging offers a non-invasive way to assess brain-specific energy metabolism. Techniques like Magnetic Resonance Spectroscopy (MRS) can measure markers such as N-acetyl-aspartate (NAA), phosphocreatine (PCr), and brain lactate levels. For example, in patients with MELAS – a mitochondrial disorder linked to psychiatric symptoms – cerebrospinal fluid lactate levels demonstrated 94.1% sensitivity and 100% specificity in identifying dysfunction.
| Biomarker Category | Specific Markers | Clinical Significance |
|---|---|---|
| Metabolic | Lactate, Pyruvate, L:P Ratio | Indicates anaerobic metabolism; L:P >20–25 suggests OXPHOS issues |
| Enzymatic | Complexes I, II, IV, Citrate Synthase | Measures ATP production; Complex I deficiency linked to depression |
| Oxidative Stress | MDA, SOD, Catalase, GSH/GSSG | Assesses oxidative damage vs. antioxidant defense |
| Mitokines | GDF-15, FGF-21 | Sensitive markers for mitochondrial stress; GDF-15 AUC up to 0.997 |
| Genetic | mtDNAcn, ccf-mtDNA | Reflects mitochondrial biogenesis and damage |
| Neuroimaging | NAA, PCr, Lactate | Brain energy metabolism markers |
These biomarkers form the foundation for more comprehensive tests that evaluate mitochondrial performance.
Lab Testing for Mitochondrial Health
Functional tests go beyond individual biomarkers, integrating multiple measures to provide a detailed picture of mitochondrial function. Respirometry, using systems like Seahorse XF or Oroboros, is considered the gold standard for analyzing oxygen consumption and cellular bioenergetics. This test requires only a small blood sample but delivers highly precise data.
“Mitochondrial oxygen consumption rate is the best measure of mitochondrial function in PBMCs.” – Theodoros Kelesidis, MD, PhD
Most tests use peripheral blood mononuclear cells (PBMCs) or platelets as stand-ins for brain tissue since these cells share key mitochondrial traits. The Mitochondrial Health Index (MHI) is a cutting-edge tool that combines respiratory chain enzyme activity with mtDNA copy numbers to evaluate mitochondrial quality. Studies show that highly stressed caregivers often have lower MHI scores, which correlate with mood disorders. These findings help tailor interventions in line with integrative psychiatric practices.
Functional testing also identifies specific enzyme deficiencies, such as reduced Complex I or Citrate Synthase activity, which can guide treatment. For example, elevated oxidative stress markers might prompt the use of N-acetylcysteine (NAC) to boost glutathione levels and counteract reactive oxygen species. Additionally, tests for calcium (Ca2+) homeostasis can reveal issues like excitotoxicity, which affects neurotransmission and is linked to mitochondrial dysfunction.
“Understanding the landscape of biomarkers in mitochondrial diseases is crucial for advancing early detection, improving patient management, and developing targeted therapies.” – PubMed
sbb-itb-23f89d4
Treatment Strategies for Mitochondrial Health
Once mitochondrial dysfunction is identified, tailored treatment strategies can help restore energy production and reduce psychiatric symptoms. These strategies include nutritional supplements, lifestyle adjustments, and emerging pharmaceutical options that tackle mitochondrial issues directly.
Nutrients and Supplements for Mitochondrial Support
Certain nutrients play a crucial role in supporting mitochondrial function and addressing psychiatric symptoms:
- CoQ10: Vital for electron transport and a strong antioxidant. Studies show that people with Major Depressive Disorder often have lower CoQ10 levels, making supplementation beneficial.
- Omega-3 Fatty Acids: They support neuronal membrane health and reduce depressive symptoms. Research links frequent fish consumption (at least twice a week) to a significant decrease in depression and suicidal thoughts.
- N-acetylcysteine (NAC): A precursor to glutathione, the body’s main antioxidant. It reduces oxidative stress and supports mitochondrial health in various psychiatric conditions. For instance, a 2018 study found that NAC improved outcomes for early-phase schizophrenia patients.
- B Vitamins: Specifically B12 and folate, these lower homocysteine levels, a neurotoxin linked to mitochondrial dysfunction and depression.
- Magnesium: A key cofactor for ATP production. Deficiency in magnesium has been associated with depressive symptoms.
- Acetyl-L-carnitine (ALCAR): Transports fatty acids into mitochondria for energy production.
- Creatine: Supports rapid ATP recycling, which is often disrupted in bipolar disorder.
| Nutrient/Supplement | Mitochondrial Role | Psychiatric Benefits |
|---|---|---|
| CoQ10 | Electron transport & antioxidant | Low levels linked to depression; boosts energy |
| Omega-3 (EPA/DHA) | Maintains membrane health | Low levels tied to depression and suicide risk |
| NAC | Glutathione precursor | Reduces oxidative stress in schizophrenia and bipolar disorder |
| Magnesium | ATP production cofactor | Deficiency linked to depressive symptoms |
| B12/Folate | Homocysteine regulation | Protects against neurotoxic effects; aids depression therapy |
| Creatine | ATP recycling | Addresses energy deficits in bipolar disorder |
Lifestyle Changes for Better Mitochondrial Function
Lifestyle adjustments can significantly improve mitochondrial health:
- Exercise: Aerobic activity promotes mitochondrial biogenesis by increasing PGC-1α, a key regulator of mitochondrial production. Exercise also boosts Brain-Derived Neurotrophic Factor (BDNF), enhancing mitochondrial respiration and energy generation.
- Diet: A Mediterranean-style diet, rich in leafy greens, healthy fats (like olive oil and avocado), and antioxidants (from foods like berries and turmeric), reduces oxidative stress and stabilizes blood sugar. For those with glucose metabolism issues, a ketogenic diet provides ketones as an alternative energy source, bypassing damaged pathways and lowering oxidative stress. Intermittent fasting (e.g., a 16:8 schedule) enhances metabolic flexibility and activates cellular repair mechanisms.
- Stress Management: Chronic stress damages mitochondrial DNA and causes fragmentation, a condition known as mitochondrial allostatic load (MAL). On the flip side, positive emotions can enhance mitochondrial energy production.
- Sleep: Quality sleep shields the brain from oxidative stress, as the brain relies solely on mitochondrial energy production due to its lack of glycogen storage.
“Mitochondria are what connect mind and body, translating social and psychological experiences into emotional responses – positive and negative – and clinically meaningful biological and physiological changes.” – Hara Estroff Marano, Editor at Large, Psychology Today
New Treatments Targeting Mitochondria
Cutting-edge treatments are now targeting mitochondrial dysfunction to improve psychiatric outcomes:
- Targeted Antioxidants: Unlike general antioxidants like Vitamin C, targeted options such as MitoQ focus on neutralizing reactive oxygen species directly within the mitochondria, offering more precise protection.
- NAD⁺ Augmentation: Nicotinamide Riboside (NR) increases NAD⁺ levels, which support mitochondrial energy metabolism and sirtuin activity (SIRT3-5). This is especially important since the brain uses 20% of the body’s energy, generating reactive oxygen species in the process.
- Mitochondrial Transplantation: This experimental therapy involves introducing healthy mitochondria into damaged cells. Research in 2024 showed its potential in restoring energy production in pediatric patients undergoing ECMO support.
- Repurposed Medications: Drugs like lithium and valproic acid enhance mitochondrial calcium signaling, inhibit cell death pathways, and improve energy production. For example, a study using iPSC-derived neurons predicted lithium responsiveness in bipolar patients with over 90% accuracy.
- Precision Psychiatry: Advances in technology, such as patient-specific iPSCs and 3D brain organoids, allow for the identification of mitochondrial biomarkers and the prediction of treatment responses.
- Drug Development: New pharmaceuticals targeting the AMPK-SIRT1-PGC-1α pathway are being developed to promote mitochondrial quality control and biogenesis.
These advancements highlight the growing potential of mitochondrial-focused treatments in addressing psychiatric conditions.
Adding Mitochondrial Health to Psychiatric Treatment
Incorporating mitochondrial health into psychiatric care involves combining functional lab testing with personalized interventions to address underlying metabolic issues. This creates a foundation for a more tailored and integrative approach to mental health treatment.
Creating Personalized Treatment Plans
Functional lab markers like TBARS, MDA, SOD, Catalase, TAS/TOS, and BDNF levels can help quantify redox balance and neuronal plasticity. These advanced tests identify disruptions in energy pathways, guiding interventions such as N-acetylcysteine, Omega-3 fatty acids, or alpha-lipoic acid. For instance, in bipolar disorder, MRS findings may direct the use of specific nutraceuticals to combat oxidative stress.
Take a patient with elevated lipid peroxidation as an example. They might benefit from Omega-3 fatty acids, which can help restore cell membrane integrity. On the other hand, someone with low antioxidant capacity may see improvements with supplements like alpha-lipoic acid or CoQ10.
Combining Mitochondrial Health with Standard Psychiatric Care
Once personalized testing is complete, integrating mitochondrial health strategies with conventional psychiatric treatments can optimize outcomes. Mitochondrial-targeted agents address issues like ATP deficits, calcium imbalances, and reactive oxygen species, complementing standard therapies. This approach is particularly beneficial for treatment-resistant patients and may even reduce side effects from certain antipsychotic medications.
“Understanding the role of mitochondrial function in psychiatric disorders may also lead to a better understanding of the therapeutic effects and side effects of current psychotropics… This could lead to strategies that might mitigate some side effects and improve our current therapies.” – Rebecca Anglin, MD, PhD, McMaster University
By supporting mitochondrial health, patients may experience enhanced cellular resilience, better neuroplasticity, and reduced oxidative damage. This approach could also address symptoms that standard treatments sometimes fail to resolve, such as the negative symptoms of schizophrenia (e.g., social withdrawal and reduced speech). Complementary methods like physical activity and cognitive stimulation further boost BDNF expression and mitochondrial respiration, creating a more well-rounded treatment plan.
When metabolic insights are combined with traditional care, clinicians can better address psychiatric symptoms in a comprehensive way.
Modyfi Approach to Mitochondrial Health
At Modyfi Health, the focus is on comprehensive functional lab testing, including nutrient panels and inflammation markers, as part of a 60-minute initial evaluation. From there, the clinic designs personalized care plans that blend medication, targeted nutritional interventions, and exercise therapy. These strategies aim to stimulate mitochondrial biogenesis through the AMPK-SIRT1-PGC-1α pathway. Continuous monitoring ensures that treatments are adjusted to meet each patient’s evolving needs.
This approach recognizes the brain’s high energy demands – it consumes about 20% of the body’s energy yet lacks glycogen storage, making it especially vulnerable to mitochondrial dysfunction. By grounding care in metabolic phenotyping, this precision psychiatry model shifts the focus from simply managing symptoms to addressing the whole person. It’s a forward-thinking way to enhance mental health care.
Conclusion: Mitochondrial Health in Modern Psychiatry
Mitochondrial health has moved from being a specialized area of study to becoming a key element in understanding psychiatric disorders. The brain’s high energy demands make it especially sensitive to even small disruptions in ATP production. When mitochondria fail to produce enough ATP, it can lead to reduced synaptic plasticity, increased oxidative stress, and weakened neuronal resilience.
The rise of precision psychiatry has allowed clinicians to use metabolic phenotyping and biomarkers – like mtDNA copy number and methylmalonic acid – to improve diagnostic accuracy and predict how patients will respond to treatments. For example, one study using iPSC-derived neurons identified lithium-responsive bipolar disorder patients with an impressive 92% accuracy rate, based solely on mitochondrial function. This level of precision paves the way for personalized treatment plans tailored to individual metabolic profiles.
Emerging evidence underscores the therapeutic potential of these biomarkers:
“Mitochondrial function biomarkers mtDNA-CN and MMA are expected to be potential therapeutic targets for depression and cognitive dysfunction, emphasizing the need for mitochondrial function monitoring and interventions in future therapies targeting neuropsychiatric disorders.” – Annals of General Psychiatry
Incorporating mitochondrial support into standard psychiatric care offers practical benefits, particularly for patients who are resistant to treatment. Combining traditional medications such as lithium or valproic acid with mitochondrial agents like N-acetylcysteine, Omega-3 fatty acids, or CoQ10 addresses both neurotransmitter imbalances and energy production deficits. This comprehensive approach not only enhances neuronal resilience but supports long-term health and vitality.
As psychiatry progresses, emphasizing mitochondrial health shifts the focus from merely managing symptoms to addressing the underlying metabolic issues. By integrating functional lab testing, lifestyle changes, and targeted nutritional therapies, this approach reflects the growing emphasis on holistic care in modern psychiatry, promoting sustainable mental health improvements.
FAQs
How does mitochondrial dysfunction impact mental health conditions like depression and bipolar disorder?
Mitochondrial dysfunction can have a profound impact on mental health by interfering with the brain’s energy supply, which is essential for its proper functioning. For example, in depression, reduced mitochondrial activity can disrupt the communication between neurons and affect mood regulation. Similarly, in bipolar disorder, mitochondrial issues may contribute to energy imbalances and increased oxidative stress, both of which are associated with mood swings and emotional instability.
Emerging research highlights the potential of targeting mitochondrial health to manage these mental health conditions. Efforts to boost energy metabolism and minimize oxidative damage are being investigated. Approaches such as strengthening antioxidant defenses and supporting mitochondrial function show promise as ways to improve mental well-being.
What lifestyle changes can help improve mitochondrial health in mental health care?
Exercise not only improves mitochondrial function but also reduces oxidative stress and increases energy production — directly easing symptoms of mental health conditions such as depression and bipolar disorder.
Diet also plays a crucial role. Eating nutrient-dense foods rich in antioxidants, B vitamins, and omega-3 fatty acids helps protect mitochondria from damage while improving brain function. Stress management techniques like mindfulness, meditation, or ensuring quality sleep further promote mitochondrial health, which ties directly to better mental well-being. Together, these habits create a well-rounded approach to caring for both your mind and body.
How are functional lab tests and biomarkers used to address mitochondrial dysfunction in mental health care?
Functional lab tests and biomarkers are essential tools for uncovering mitochondrial dysfunction in mental health care. These tests focus on areas like energy production, oxidative stress, and mitochondrial DNA health – factors often associated with psychiatric conditions such as depression and bipolar disorder.
The brain’s high energy demands mean that even small shifts in cellular health can change how you feel, think, and perform. At Modyfi Health, we move beyond just managing symptoms by investigating biological root causes through precision psychiatry and functional testing.
If you’re ready to design a health blueprint that actually fuels your brain, we invite you to book a Discovery Session and explore your path to lasting wellness.
